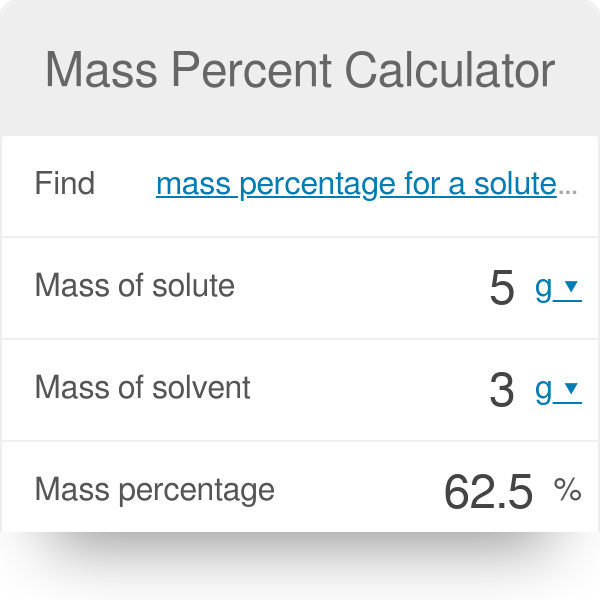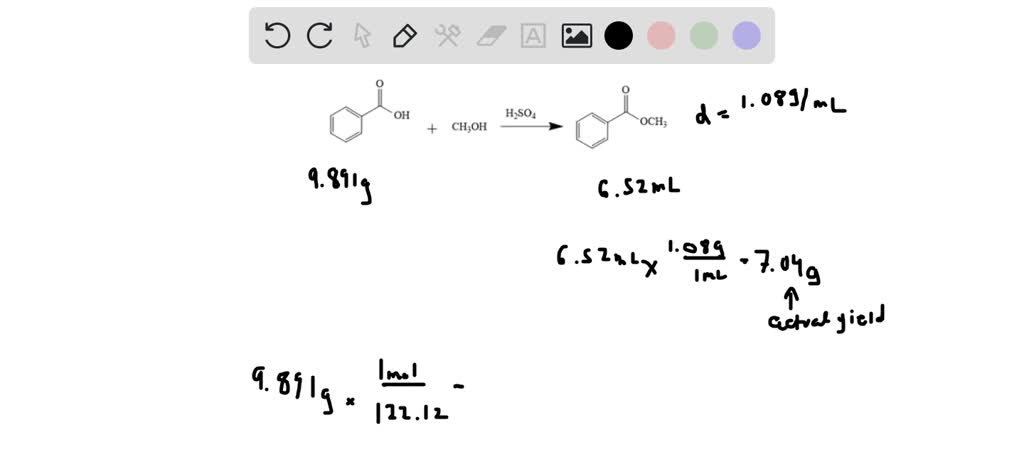
SOLVED: Take the mass of benzoic acid used and the yield of methyl benzoate. Use the yield to calculate your percent yield and theoretical yield. Benzoic acid; 9.891g , Methyl Benzoate; 6.52 mL

Percent Composition: The percent by mass on an element in a compound Is the number of grams of the element in a formula divided by the total mass of the. - ppt

Question Video: Calculating the Percent by Mass of the Active Ingredient in an Anti-inflammatory Ointment | Nagwa

Baker's Percentage Method and Calculator | Crafty Baking | Formerly Baking911 | CraftyBaking | Formerly Baking911



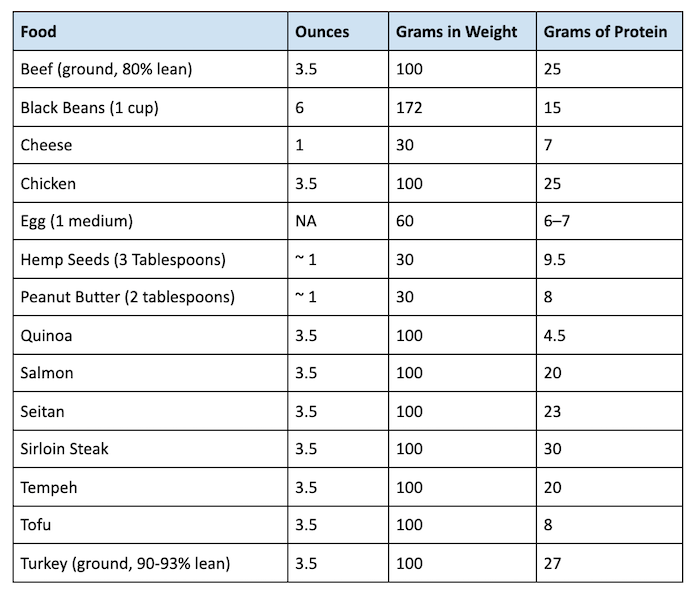


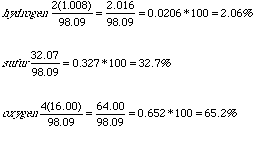






:max_bytes(150000):strip_icc()/mass-percent-composition-example-609567_V2-01-89c18a9d30ea43b494d09b81f7ffefc1.png)