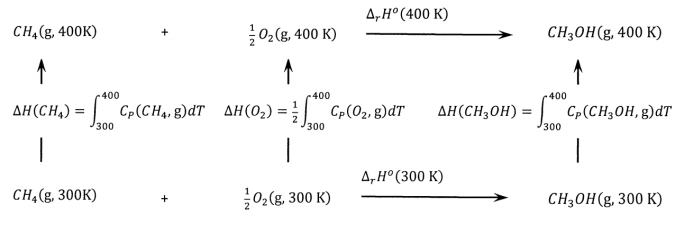
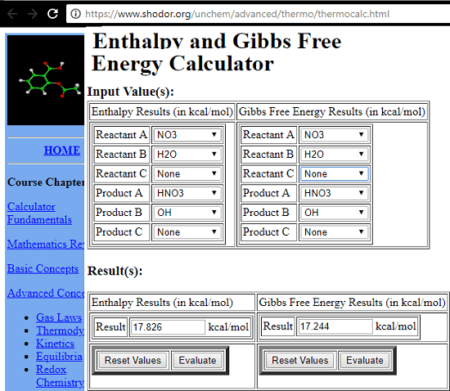
Question Video: Calculating Standard Enthalpy of Combustion of Methane Using Standard Enthalpies of Formation of Methane and Carbon Dioxide | Nagwa

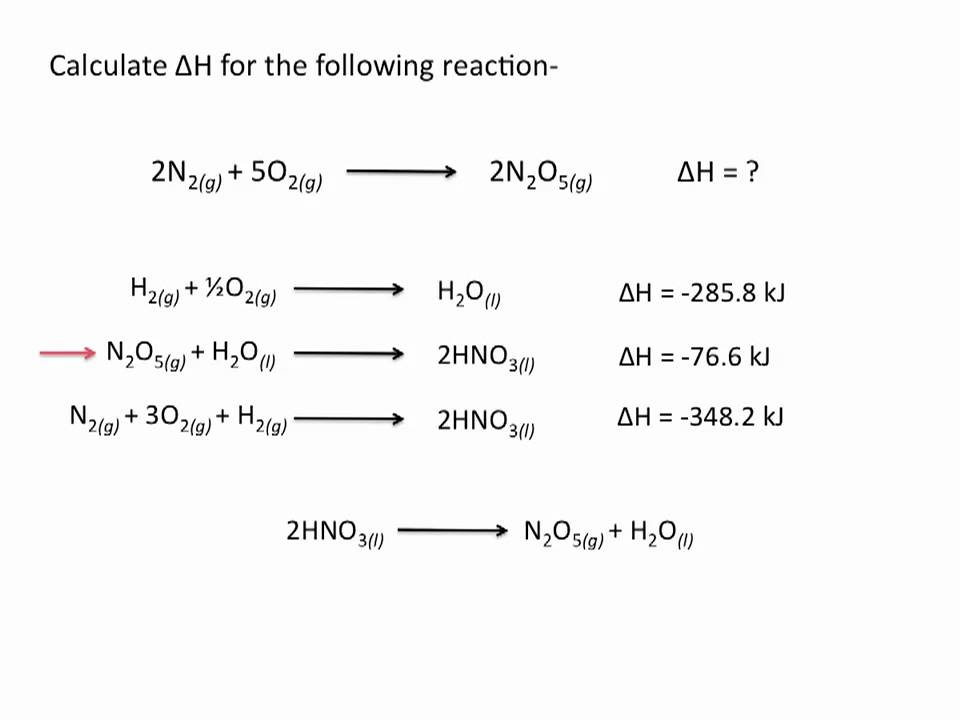
Calculate ΔH/kJ for the following reaction using the listed standard enthalpy of reaction data. 2N2 (g) + 5O2 (g) ⟶ 2N2O5 (s) N2 (g) + 3O2 (g) + H2 (g) ⟶ 2HNO3 (
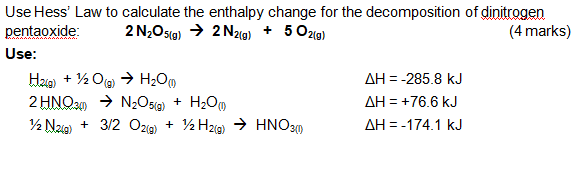
Use Hess' Law to calculate the enthalpy change for the decomposition of dinitrogen pentaoxide? | Socratic

3:04 calculate the molar enthalpy change (ΔH) from the heat energy change, Q - TutorMyself Chemistry

Calculate the standard enthalpy of reaction for the following reaction using the listed enthalpies of reaction : 3Co(s)+2O(2)(g)rarrCo(3)O(4)(s) 2 Co(s)+O(2)(g)rarr2CoO(s)," "DeltaH(1)^(@)=-475.8 kJ 6CoO(s)+O(2)(g)rarr2Co(3)O(4)(s),DeltaH(2)^(@)=-355.0 kJ